Digital Pills
Digital pills, also known as smart pills or ingestible sensors, are a category of small devices that can safely travel throughout the body while communicating with an external device.[1] Currently, the application of digital pills can enable physicians to image internal structures, measure critical values, and monitor patient activity without invasive procedures.[2] The use of digital pills also raises important ethical considerations that affect patients, physicians, and society as a whole.[2]
Contents
Background
One of the earliest inventions for ingestible sensors comes in 1957 from the work of Jacobson and Mackay who wanted to use radio frequency transmission to get real-time reports of different vital measurements from patients.[2] Jacobson and Mackay called their device endoradiosonde and it was able to measure pH, temperature, and pressure in the gastrointestinal tract.[4] The next major breakthrough came in the 2000s when SmartPill, a wireless motility capsule (WMC), was developed, approved, and sold across the United States to measure pH, pressure, and temperature all at once for a more accurate diagnosis of gastric emptying.[4] In 2001, a video capsule endoscope (VCE) was also approved by the Food and Drug Administration for widespread commercial use.[4] Most recently, the FDA approved another digital pill, Abilify MyCite, which can be used to monitor patient adherence to their medication.[5] The 2000s introduced the miniaturization of electronic circuits and batteries, advancements in biomaterials, and innovation in drug developments which together have paved the way for digital medicine to progress.[4] Currently, digital pills exist or are being tested for a wide range of applications including temperature sensing[6], pH monitoring[7], motility sensing[8], capsule endoscopy[4], biopsy[4], gas sensing[4], inflammation detection[4], drug delivery[4], and adherence monitoring[4]. Almost all digital pills require three components for their utilization: 1) the capsule with a sensor; 2) receiver usually worn by the patient; and 3) a computer or mobile application with compatible software that can be used to store and analyze the data[2].
Types of Smart Pills
Imaging Capsules
These smart pills are a type of diagnostic instrument that enables doctors to collect visual data about the structural integrity and condition of organ lining inside the different organs of the gastrointestinal (digestive) tract.[2] A major advantage of imaging capsules is that they provide an alternative to traditional imaging techniques that use tube endoscopes which are more invasive.[2] Endoscopy instruments also tend to be large and rigid which limits access to many areas of the gastrointestinal tract and brings greater discomfort to patients.[2] A Typical wireless imaging capsule systems have 3 components: 1) capsule endoscope; 2) sensing system such as a pad/patch or belt attached to the patient; and 3) some sort of computer or application on a mobile device with the right software to receive the information.[9] Over the years, imaging capsules have made great progress with improved algorithms for detecting hemorrhage and lesions and enhanced video quality.[2] Due to the uniqueness of each organ within the gastrointestinal tract, different capsules with specific features were developed for each organ.[2]
Current Products

PillCam SB by Given Imaging Inc: This is the first swallowable camera capsule released by Given Imaging inc. and it is used to analyze the small intestine (also called small bowel, this is where SB comes from).[2] Since the initial release in 2000, newer versions have been released with greater battery power and imaging resolution.[2] The capsule consists of a video camera, light source, batteries, radio transmitter, and an antenna.[11] The capsule is able to take 50,000 pictures over the course of 8 hours which averages to about two pictures every second.[11] The images taken by the capsule are transmitted to a belt worn by the patient which can relay the information to a workstation or other electronic device with the appropriate software.[11] To prepare for the capsule, patients are required to fast for 10 hours and then are given the pill with water to swallow; once the capsule has entered the gastrointestinal tract it will capture images for about eight hours and the patient can resume normal daily activities right after consumption.[11]
PillCam ESO by Given Imaging Inc: This capsule is used to take images of the esophagus and there are two cameras, one at each end, to get more information in less time.[2] The pill travels down the esophagus very quickly and as a result, the pill is designed to have a short battery life but a high frame rate.[2], [9] Patients are asked to fast two hours before their examination and when they come in they are fitted with 3 thoracic sensors that are connected to a data recorder on some type of workstation with the appropriate software.[9] The patient is given 100mL of water while standing and then they ingest the activated capsule with another 10mL of water that they drink while laying down on their back.[9] There is a 5 minute ingestion period which consists of 2 minutes with the patients laying down on their back (supine position), 2 minutes with the patient raised to 30 degrees, and then an additional minute of the patient raised to 60 degrees.[9] Lastly, the patient remains seated in an upright manner for 15 minutes to maximize the time for the capsule to capture images as it travels through the esophagus.[9]
There are many other ingestible sensors available on the market such as PillCam Colon, Endo Capsule, OMOM capsule, and MiroCam capsule that are specific to other organs of the gastrointestinal tract for optimized imaging.[2]
Temperature Sensing Capsules
Temperature sensing smart pills are a diagnostic tool that can be used to make sure body temperature remains within the homeostatic range and they can also be used to measure heat stress in patients.[2] There is a growing trend of the inclusion of temperature sensing capabilities in other pills that have a different primary function.[2]
Current Products
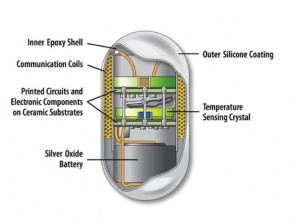
CorTemp Ingestible Sensor: The capsule consists of a thermistor-based temperature sensor and is usually used for core body temperature measurements.[2] The capsule is coated in silicone and medical epoxy and contains a micro battery, quartz crystal, communication coil, and a circuit board.[6] Once a patient ingests the sensor, the crystal within it vibrates at a frequency relative to the body’s internal temperature which produces a magnetic flux and consequently transmits a low-frequency signal to a data recorder device.[6] The recorder device converts the signals from the sensor to a digital format and also has the ability to store data which can later be transferred to a computer for more in-depth analysis.[6] The sensor capsule will naturally pass through the patient’s gastrointestinal tract within 24 to 36 hours.[6]
pH Monitoring Capsules
pH monitoring capsules serve as a diagnostic measure that can help physicians track pH fluxes in patients.[2] pH is an important characteristic that measures the level of acidity in a given environment. This characteristic becomes important when working with the gastrointestinal tract because different organs maintain specific local pHs. Imbalances in pH can lead to many diseases such as inflammatory bowel disease[12] or gastroesophageal reflux disease (GERD)[7] and monitoring for these changes can be life-saving.
Current Products
Bravo pH monitoring system: The Bravo system has 3 components: 1) wireless capsule; 2) recording device worn by the patient; and 3) a software system available for computers that properly manages all the data and allows for analysis.[13] Unlike traditional pills, the Bravo pH sensor must be temporarily inserted into the patient’s esophagus during an endoscopy procedure.[13] Patients are asked to fast 4-6 hours before their appointment and when they arrive a local anesthetic will be applied to the back of their throat.[13] The entire procedure of the upper endoscopy to place the capsule will take approximately 5 minutes.[13] This system can be used to actively monitor acid-base levels within the esophagus and stomach to treat acid reflux and GERD.[13]
Medication Monitoring Pills
The medication monitoring pills can be used by patients and doctors to make sure that the patients are taking their pills according to their prescription and that they are not skipping any of their medications.[5] Medication non-adherence is a growing problem that is preventable and the monitoring pills provide a viable option.[5] Medication non-adherence can often result in serious consequences, especially for chronic diseases such as hypertension and diabetes where an estimated 40-50% of patients are believed to be affected by medication nonadherence.[14] This likely contributes to 100,000 preventable deaths and $100 billion dollars in preventable medical expenses each year.[14] Tracking patient activity, digital pills that monitor consumption aim to resolve medication non-adherence and provide physicians with more accurate information regarding the effectiveness of treatments.[14]
Current Products
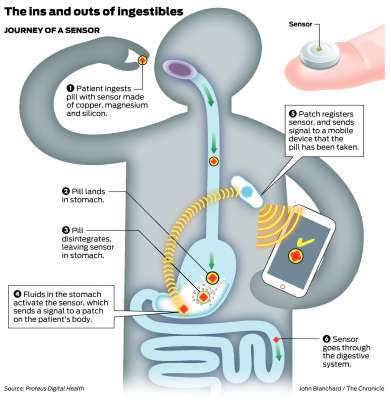
Abilify MyCite: The Abilify MyCite digital pill was developed by Otsuka Pharmaceutical Co. and it was approved by the FDA in 2017.[5] The actual drug itself is Abilify, also known as aripiprazole (generic name), and it is used in the treatment of schizophrenia and bipolar disorder.[15] The MyCite digital pill sensor was developed by Proteus Digital Health and it is composed of a silicon chip with a logic circuit and two metals (magnesium and copper).[15] When the pill comes into contact with solutions consisting of polar molecules (like gastric acid in the stomach) it dissolves leaving behind the silicon chip.[5], [15] When the chip interacts with the surrounding solution it becomes activated and is able to transmit a transient electrical signal (modulated current) to a wearable patch on the patient.[15] The signal received by the patch consists of a single number which informs the patch what medication has been ingested.[15] Through Bluetooth, the wearable patch can relay the information to mobile devices on a compatible app.[5], [15] This information, with the required patient consent, can be shared with the patient’s loved ones, caretakers, and/or physicians to make sure that the patient avoids medication non-adherence.[5]
Capsules in Development
Research into the field of smart pills continues to grow as new illnesses are discovered and new therapeutics are needed.[2] Currently, research on gas sensing capsules, visible and infrared wavelength spectrometry capsules, Raman Spectroscopy, Confocal Microscopy and Optical Coherence Tomography, and Electrochemical sensing capsules is being done.[2] Some of these pills are further in clinical trials with human patients, others are still in animal trials, and some are still just ideas.[2]
Ethical Concerns
Patient-Related
These are ethical concerns that arise from smart pills that primarily affect patients.
Autonomy and Informed Consent
Typical medical procedures, from simple tasks such as measuring a patient’s blood pressure to more invasive tasks such as drawing blood all require informed consent from the patient.[16] The addition of technology brings in an additional stipulation, the user agreement.[16] These forms tend to be extensively long and written by lawyers to describe in detail how the software and hardware of the product can be used.[16] This can lead to issues in informed consent as the patients may not always fully understand the user agreement.[16] Traditional informed consent gives the patients the convenience of a face-to-face conversation with their doctor to address all their concerns and obtain a proper understanding of the situation.[16] User agreements do not offer such services and many times patients do not fully realize what they are agreeing to due to comprehension difficulties of the form and this can potentially threaten patient autonomy.[17] User Agreement forms can also be updated at any time without notice and are non-negotiable which may infringe on patient autonomy.[16] Many user agreement forms require the patients to allow the company to collect data so that they can continue to improve their product.[16] If a patient is uncomfortable, their alternative is to find a completely new treatment option that may not be feasible for them.[16] This can potentially put the patient in an uncomfortable situation where they feel as if they do not have complete control over their choices.[16] Since doctors can monitor patients from afar, digital pills may allow patients to remain in their homes longer without the need for constant supervision increasing their autonomy as well.[18] Digital pills appear to play a double role, restricting and enhancing patient autonomy in different manners.[18]
Data Management
Due to the relative novelty of digital pills and digital medicine, there are no experienced guidelines set in place to protect the patients, physicians, researchers, or other parties involved from unethical practices.[16] This can raise potential issues regarding the data and use of sensitive patient information.[16] Digital medicine is unique because it gives patients an additional responsibility of deciding who they want to share their information with, whether this is family, researchers, physicians, or others.[16] Current guidelines do not, however, always give patients the control to decide how their information is used.[16] The power over data usage is based on several factors, but not limited to the presence of patient identifiers, the manner in which the data is collected, and if the data was collected as part of a research study.[16]
Confidentiality of Private Data
Another pressing issue that is likely to be the subject of debate is the preservation of the confidentiality of private data about patients.[16] Traditionally, patient data is only shared with the patient and required health professionals involved in the treatment or care of the patient who have proper training and experience in dealing with sensitive information.[16] With digital medicine this exclusive group can suddenly expand to include many more parties such as relatives, friends, roommates, and even insurance companies or software developers.[16] While doctors and other health professionals know their responsibility and duty to their patients, the other parties do not.[17] Friends or family members involved in the daily life of the patient who have access to such sensitive information are not under the same obligations as members of the medical community to keep the data confidential.[17] This raises an ethical concern of what the rules and responsibilities should be for those included in maintaining the confidentiality of a patient’s data.[16]
Provider-Related
The implementation of the smart pills will also affect the physician and their responsibilities towards patients.
Patient Trust and Expectations
With digital pills, a physician can now monitor a patient even when they are outside of the clinic. This significantly alters the conventional system of trust between patients and physicians.[17] Before digital pills, patients self-reported whether they were following their prescription, and if the treatment was not working the physician had no way of figuring out whether the problem was a case of medication nonadherence or an ineffective treatment.[16] Digital pills can resolve this difficulty as medication ingestion can be monitored.[16] This shifts the framework of providing care from patients voluntarily providing the physician with information to physicians having constant access to patient information[17]. Another concern that arises is the mutual understanding of expectations.[16] A physician prescribing digital pills to numerous patients will not have the time to closely monitor each individual patient.[16] However, patients receiving the digital pills are likely to assume that their physician is closely monitoring them, or else they would not have switched to this medication.[16] Misalignment of expectations can lead to serious consequences if patients become over-reliant on physicians monitoring them from a distance and they start spacing their appointments further apart and visit their clinicians less in person.[18] Ethical issues may arise if patients decide to take legal actions following health complications because a physician may not have been monitoring their health records as closely as the patient would have preferred.[16]
Physician-Patient Relationship
Digital pills will change the dynamic of the physician-patient relationship.[19] The autonomy given to patients by digital pills comes at the cost of creating a more distant relationship with physicians since they will no longer see them as frequently.[19] The relationship between patients and physicians will also become much more transparent as physicians will get daily updates on the condition of their patients and patients will get feedback from physicians when necessary based on their reports.[19] With these changes in the dynamic, ethical questions may arise on the issue of how these new factors will influence the ability of doctors to provide the best care to their patients.[19]
Physician Autonomy
The utilization of digital pills is likely to limit the autonomy of physicians who prescribe them.[16] Medical research companies and pharmaceuticals who overlook the products will have access to data not only about who is prescribed the medication but also on how often a clinician logs into their system to check on the patients.[16] This will allow other parties to oversee not only how often a physician checks a patient’s records but also for how long.[16] Such a level of close supervision really hinders the professional autonomy of doctors and may have ethical implications if such measures are used to evaluate physicians by insurance companies.[16] These implications could affect how a physician decides to create a treatment plan for a diagnosis which could have ethical considerations as well.[16]
Societal-Related
The implementation of smart pills will influence healthcare beyond just the patient and physician.
Insurnace Companies
The cost of digital pills tends to be much greater than traditional medicine that can ultimately fulfill the same purpose.[16] For example, the Abilify digital pill is double the cost of the original version without the digital software.[16] With such expenses, it is probable that insurance companies will likely require evidence and even access to certain metrics to make sure that the digital medications really are worth the extra money.[16] This may come in the form of access to patient data regarding adherence to prescriptions or even requiring clinicians to check on patients a certain number of times.[16] Such demands will come at the cost of both patient and physician autonomy.[16]
Equitable Access
A requirement of digital pills is having access to a compatible device such as a smartphone.[16] According to the Pew Research Forum, 77% of U.S. adults have a smartphone,[20] however, when analyzing income level this value drops to 64% when looking at adults making under $30,000 a year.[21] With access to compatible devices being dependent on socioeconomic situations, there is likely to be unequal access to digital pills unless some sort of solution or aid is given to those who do not have access to compatible devices.[16]
See Also
References
- ↑ Farr, C. (2017, December 18). The first 'Digital Pill' has just been approved - here's how it could revolutionize health care. CNBC. Retrieved February 11, 2022, from https://www.cnbc.com/2017/11/14/what-is-a-digital-pill.html.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 2.20 Kalantar-zadeh, K., Ha, N., Ou, J. Z., & Berean, K. J. (2017). Ingestible sensors. ACS Sensors, 2(4), 468–483. https://doi.org/10.1021/acssensors.7b00045.
- ↑ Thadani, T. (2017, June 13). Did you take your pill? ingestible sensors can tell. San Francisco Chronicle. Retrieved February 11, 2022, from https://www.sfchronicle.com/business/article/Did-you-take-your-pill-Ingestible-sensors-can-11206980.php
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 Mau, M. M., Sarker, S., & Terry, B. S. (2021). Ingestible devices for long-term gastrointestinal residency: A Review. Progress in Biomedical Engineering, 3(4), 042001. https://doi.org/10.1088/2516-1091/ac1731.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 impacX Team. (2021, May 10). Smart pills: The era of Edible Digital Pills has arrived! impacX. Retrieved January 28, 2022, from https://impacx.io/blog/smart-pills-what-are-these-edible-digital-pills/.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Cortemp. HQ, Inc. (2022, January 24). Retrieved February 11, 2022, from https://www.hqinc.net/cortemp/#:~:text=Our%20CorTemp%C2%AE%20Ingestible%20Core,the%20outside%20of%20the%20body.
- ↑ 7.0 7.1 Mayo Foundation for Medical Education and Research. (2020, May 22). Gastroesophageal reflux disease (GERD). Mayo Clinic. Retrieved February 11, 2022, from https://www.mayoclinic.org/diseases-conditions/gerd/symptoms-causes/syc-20361940.
- ↑ Farrar, J. T., Zworykin, V. K., & Baum, J. (1957). Pressure-sensitive telemetering capsule for study of gastrointestinal motility. Science, 126(3280), 975–976. https://doi.org/10.1126/science.126.3280.975.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Wang, A., Banerjee, S., Barth, B. A., Bhat, Y. M., Chauhan, S., Gottlieb, K. T., Konda, V., Maple, J. T., Murad, F., Pfau, P. R., Pleskow, D. K., Siddiqui, U. D., Tokar, J. L., & Rodriguez, S. A. (2013). Wireless capsule endoscopy. Gastrointestinal Endoscopy, 78(6), 805–815. https://doi.org/10.1016/j.gie.2013.06.026
- ↑ Given Imaging. (n.d.). PillCam® SB - digestive endoscopy endoscopy capsule by given imaging: Medicalexpo. Medical Expo. Retrieved February 11, 2022, from https://www.medicalexpo.com/prod/given-imaging/product-75056-469484.html
- ↑ 11.0 11.1 11.2 11.3 John Hopkins Medicine. (n.d.). PILLCAM™ SB FREQUENTLY ASKED QUESTIONS (FAQ). John Hopkins Bayview Medical Center. Retrieved February 11, 2022, from https://www.hopkinsmedicine.org/johns_hopkins_bayview/_docs/medical_services/gastroenterology/pill_cam_sb_faqs.pdf.
- ↑ NUGENT, S. G. (2001). Intestinal luminal ph in inflammatory bowel disease: Possible determinants and implications for therapy with aminosalicylates and other drugs. Gut, 48(4), 571–577. https://doi.org/10.1136/gut.48.4.571.
- ↑ 13.0 13.1 13.2 13.3 13.4 Bravo Ph test: What it is, how it's done & risks. Cleveland Clinic. (n.d.). Retrieved February 11, 2022, from https://my.clevelandclinic.org/health/diagnostics/12042-esophagus-48-hour-bravo-esophageal-ph-test.
- ↑ 14.0 14.1 14.2 Kleinsinger, F. (2018). The unmet challenge of medication nonadherence. The Permanente Journal. https://doi.org/10.7812/tpp/18-033.
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 Emspak, J. (2017, November 16). FDA approves First 'Digital' pill: How does it work? LiveScience. Retrieved January 28, 2022, from https://www.livescience.com/60963-how-does-digital-pill-work-abilify.html.
- ↑ 16.00 16.01 16.02 16.03 16.04 16.05 16.06 16.07 16.08 16.09 16.10 16.11 16.12 16.13 16.14 16.15 16.16 16.17 16.18 16.19 16.20 16.21 16.22 16.23 16.24 16.25 16.26 16.27 16.28 16.29 16.30 16.31 16.32 16.33 16.34 16.35 Klugman, C. M., Dunn, L. B., Schwartz, J., & Cohen, I. G. (2018). The Ethics of Smart Pills and self-acting devices: Autonomy, truth-telling, and trust at the dawn of Digital Medicine. The American Journal of Bioethics, 18(9), 38–47. https://doi.org/10.1080/15265161.2018.1498933.
- ↑ 17.0 17.1 17.2 17.3 17.4 de Miguel Beriain, I., & Morla González, M. (2020). ‘digital pills’ for mental diseases: An ethical and social analysis of the issues behind the concept. Journal of Law and the Biosciences, 7(1). https://doi.org/10.1093/jlb/lsaa040.
- ↑ 18.0 18.1 18.2 Digital Pills Raise informed consent, privacy concerns. Relias Media - Continuing Medical Education Publishing. (n.d.). Retrieved February 11, 2022, from https://www.reliasmedia.com/articles/139823-digital-pills-raise-informed-consent-privacy-concerns.
- ↑ 19.0 19.1 19.2 19.3 Slabodkin, G. (2021, September 28). Digital Pills come under fire for privacy, patient-provider issuesGreg. Health Data Management. Retrieved February 11, 2022, from https://www.healthdatamanagement.com/articles/digital-pills-come-under-fire-for-privacy-patient-provider-issues.
- ↑ Smith, A. (2020, August 25). Record shares of Americans have smartphones, Home Broadband. Pew Research Center. Retrieved February 11, 2022, from https://www.pewresearch.org/fact-tank/2017/01/12/evolution-of-technology/.
- ↑ Pew Research Center. (2021, November 23). Demographics of mobile device ownership and adoption in the United States. Pew Research Center: Internet, Science & Tech. Retrieved February 11, 2022, from https://www.pewresearch.org/internet/fact-sheet/mobile/.
| ←Back • ↑Top of Page |